
Instructions for the reader:
The text contains elements of the little experience I have. It can, therefore, be perfectly questionable, especially the units and data that emerged from literature.
All ventilation settings are under medical supervision and approval (for lovers of nursing diagnosis, this is a collaborative problem). Since certain aspects are strongly linked to local resources and “attitudes”, the interface types and ventilator models will not be discussed.
In recent decades the efficiency of NIV (Noninvasive ventilation) has been used for several clinical conditions, such as cardiogenic pulmonary edema, exacerbation of the COPD, and acute respiratory failure in immunocompromised patients.
Difficult cases can also be treated with NIV, thanks to the growing experience of professionals in the field. My first approach with the patient is showing the various principles and materials, and briefly explaining how they work. I try to reassure the patient about the process in order to make him/her comfortable and encourage him/her to cooperate. It is usually very helpful to establish a “trust contract” with specific and simple goals to achieve in a short period of time. Before the expiration date, I instruct the patient to coordinate his breathing with the cycles of the ventilator. To reduce stress and improve comfort, I position the client on the bed with supportive devices such as pillows and rolls and raise the head of the bed to 60-90° degrees. I position the interface on the patient’s face and before I ensure it, I make sure to keep it in that position for one or two minutes with the ventilator support (see setting supports at the end of this article). I adjust the mask when it fits comfortably and when there is no seal tightness (initially, just enough to avoid excessive losses). I ask the patient at the initial stages to close his/her eyes, in order to relax as much as possible and to take a “nasal breath” when the air hunger decreases. I avoid the recurring and devastating phrase “please try to breathe well” (if the person can breathe well, he/she would not need to undergo NIV!).
Individuals should be managed without sedation, but if it is necessary, the next recommended step is to prevent alveolar hypoventilation and the loss of airway protection.
Initial ventilator settings are critical to ensure a good balance between tolerance of NIV and a proper and better gas exchange.
Inspiratory pressure: although the clinical conditions that require the use of NIV are multiple, one of the fundamental objectives is to rest inspiratory muscles. It is recommended to choose inspiratory support (PS or IPAP) to obtain an exhaled tidal volume of 6-8 ml/kg (ideal weight). Usually, this goal is easily accomplished within the first 5 to 10 minutes, starting with low support (3-5 cmH2O), with a subsequent increase of 2-3 cmH2O at a time, with collateral assessments of air leaks (< 0,4 L / s o < 25 L / min) and degree of the patient’s tolerance. This process can give the patients time to familiarize themselves with the treatment and allow the responses to be evaluated. (Figure 1).
The support is generally adjusted to make the inspiratory flow in a decreasing manner. As far as positive end-expiratory pressure is concerned, it follows the steps previously mentioned. I start cautiously from 3-5 cmH2O up to the target. Summative assessments of applied airway pressures require subsequence adjustments of PS and/or PEEP observing for each variable to affect patient-ventilator interaction. It is often recommended to use higher clinically indicated settings and the patient’s tolerated PEEP & PS levels to increase the elastance and improve trigger sensitivity when using a helmet. As a general rule, it is advised to carry out single variations of the setting spaced out by a reasonable amount of time to evaluate the response obtained from the patient-ventilator interaction.
The fraction of inspired oxygen (FiO2) is usually titrated to obtain SpO2>90%.
The inspiratory trigger is primarily flow-based and is set as sensitive as possible as long as it does not generate auto-cycling.
The transition of the ventilator from inspiration to expiration (cycling) should occur in response to the end of neural inspiratory effort. An initial setting of 30 to 50% is recommended for obstructive pulmonary disease and approximately 10 to 20% for other types of acute respiratory failure. It is strongly evidenced that slower cycling may increase inspiratory time, while faster cycling in COPD subjects is generally beneficial to reduce the risk of delayed cycling, intrinsic PEEP, and ineffective effort. What was mentioned in the latter can be applied only if the inspiratory activity is not excessive, and if contrary, the possible setting variations are almost uninfluential.
Rise time is important to reduce inspiratory effort and improve synchronization. The setting, however, must be adapted to the patient’s tolerance and comfort. An initial rise time of 0.10 seconds can be adapted to most cases. Ventilator alarms must be well set to maintain safety and reduce noise pollution (in the first few minutes I silence all the alarms). Next, I set the apnea alarm, low or high tidal volume, minute ventilation minimum, respiratory rate (generally more than 30 or adapted to the respiratory drive), peak pressure (30-35 cmH2O), and PEEP (1cmH2O lower than the set value).
From the beginning stage, it is important to monitor the patient regularly every 10 to 15 minutes for the first two hours. Monitoring is essential to determine if the initial goals have been achieved and the frequency of the subsequent evaluation will depend on the evolution of the clinical results of the patient. When there is little improvement or further deterioration, the patient will need more frequent assessments. A key goal is dyspnea alleviation, which depends on adequate support and the person’s ability to relax enough for the ventilator to take on some of the respiratory workloads.
Monitor for these objective results: respiratory and heart rate, blood pressure, level of consciousness, use of accessory respiratory muscles, and skin breakdown. Proper ventilator settings will reduce dyspnea, tachypnea and accessory muscle use. Inspiratory accessory muscle activation (sternocleidomastoid and scalene muscle) usually indicates insufficient support, while respiratory accessory muscle activation messages inappropriate cycling or PEEP level settings. After unloading the respiratory muscles from the excessive work, I begin to reduce the support by accepting a level of respiratory activity that can be compatible with the muscular resources. Control of expiratory volume is important to ensure that adequate alveolar ventilation wears high volume (> 9,5 ml/Kg Ideal weight) in subjects with moderate to severe hypoxemia, which has been associated with NIV failure. I evaluate the patient-ventilator asynchronies and air leaks through the analysis of the pressure/flow/volume waveforms. The asynchronies must be managed by reducing air losses by adjusting the inspiratory trigger, pressure support levels, cycling, and rise time.
Evaluation of respiratory exchanges is suggested with blood gas analysis before treatment, as well as 30 to 60 minutes after the treatment’s initiation. Successful NIV treatment is assessed by SpO2, pH e PaCO2, within one to two hours of initiating ventilatory support.
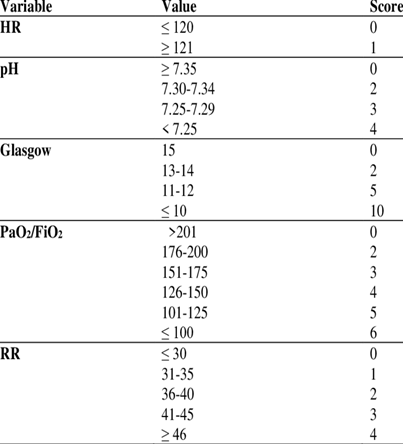
I recommend a quick evaluation using the HACOR score, figure 2, (although it needs further evaluation studies, it is a potentially useful tool for predicting an NIV treatment failure in hypoxemic patients for >5 scores). The success of the NIV treatment does not depend solely on respiratory insufficiency, but on the: characteristics of the subjects; moment in which the treatment is started; ventilator settings; degree of experience; interdisciplinary collaboration of the healthcare professionals. In Chart #1, I summarized the main nursing topics according to each case.
| Case | Nursing arrangements |
| Subject does not improve within 1 or 2 hours | Prepare the intubation materials. Intubation delay increases the risk of respiratory or cardiac complication with related morbidity and mortality. |
| Pressure sore prevention | Place silicones or hydrocolloid seals in the sites of increased pressure. Consider the use of a nasal mask; ensure treatment breaks of 30 to 90 minutes every 3-4 hours. Avoid overtightening of the fixation system. |
| Claustrophobia | Consider the use of a nasal mask. |
| Over productive cough | Consider the use of nasal mask. |
| Airway humidification | Active humidification could reduce the work of breathing and carbon dioxide accumulation compared to heat and moisture exchangers by reducing dead space and flow resistance. |
| Gastric distension | It is rarely clinically significant as long as airway pressures remains below esophageal opening pressure (PS + PEEP ≤ 18 cmH2O). Possible complications are vomiting and bronco aspiration. Routine use of a nasogastric tube is not recommended because it promotes air leaks and increases the risk of cutaneous lesions. |
| Undergoing sedation | Monitor for alveolar ventilation or loss of airway protection. |
| Respiratory muscles activation | Monitor the inspiratory accessory muscle activation (sternocleidomastoid and scalene muscles) and expiratory muscle activation. |
| Objective evaluation | Respiratory and heart rate, blood pressure, level of consciousness, use of accessory muscles, reduction/absence of asynchronies, blood gas analysis evaluation, and cutaneous lesions. Consider HACOR score. |
| Subjective | Reduction or absence of dyspnea and patient’s comfort. |
Thank you,
Cristian
English version edited by Giulia Azzini
Consulted bibliography:
- Davidson AC, Banham S, Elliott M, et al. BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax 2016;71:ii1–ii35.
- Rochwerg B, Brochard L, Elliott MW, et al. Official ERS/ATS clinical practice guidelines: noninvasive ventilation for acute respiratory failure. Eur Respir J 2017;50.
- Bello G, De Pascale G, Antonelli M. Noninvasive ventilation: practical advice. Curr Opin Crit Care 2013;19:1-8.
- Nava S, Hill N. Non-invasive ventilation in acute respiratory failure. Lancet 2009;374: 250–9.
- Jaber S, Chanques G, Matecki S, et al. Comparison of the effects of heat and moisture exchangers and heated humidifiers on ventilation and gas exchange during non-invasive ventilation. Intensive Care Med 2002;28:1590-4. doi: 10.1007/s00134-002-1441-0.
- Vargas F, Thille A, Lyazidi A, et al. Helmet with specific settings versus facemask for noninvasive ventilation. Crit Care Med 2009;37:1921-1928.
- Carteaux G, Millán Guilarte T, De Prost N, et al. Failure of noninvasive ventilation for de novo acute hypoxemic respiratory failure: role of tidal volume. Crit Care Med. 2015;44:282-290.
- Duan J, Han X, Bai L, et al. Assessment of heart rate, acidosis, consciousness, oxygenation, and respiratory rate to predict noninvasive ventilation failure in hypoxemic patients. Intensive Care Med. 2017;43:192-199.